TLDR¶
• Core Points: A novel AGCL powder, created by a multidisciplinary team including a South Korean Army Major, rapidly seals severe wounds, offering a potential critical tool for battlefield and civilian trauma care.
• Main Content: AGCL powder’s design blends materials science and bioengineering to form a fast-acting, adhesive sealant that promotes rapid hemostasis and wound stabilization.
• Key Insights: The spray-on approach aims to be easy to deploy, cost-effective, and compatible with various wound types, but requires further validation in clinical settings.
• Considerations: Safety, biocompatibility, manufacturing scalability, regulatory approval, and real-world effectiveness under diverse conditions must be established.
• Recommended Actions: Pursue phased clinical trials, optimize formulation for broad use, and develop training protocols for first responders and medical personnel.
Content Overview¶
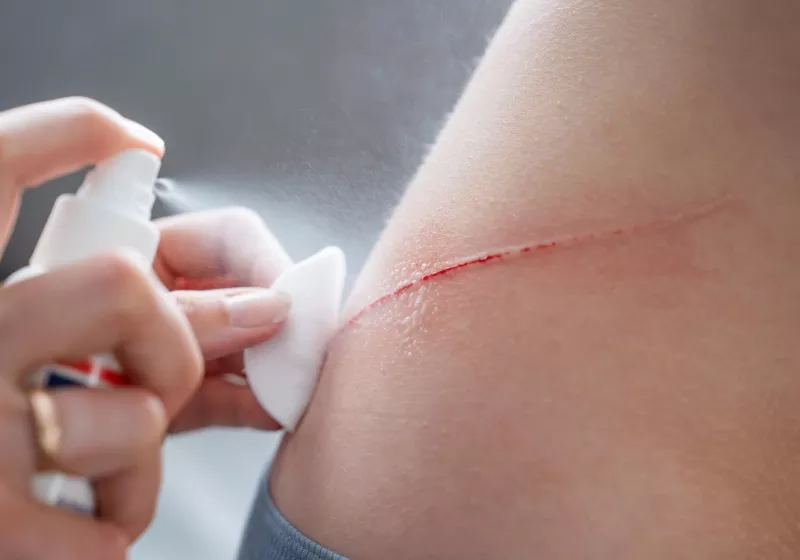
In recent developments at the intersection of materials science and bioengineering, researchers introduced a spray-on powder designed to rapidly seal life-threatening wounds. The agent, referred to as AGCL powder, emerged from a cross-disciplinary collaboration that integrated expertise in materials science with biological engineering. An active-duty South Korean Army Major, whose battlefield experiences provided practical grounding for the tool’s real-world application, contributed as a key participant in shaping the design priorities. The project reflects a broader trend toward field-ready trauma solutions that can be deployed quickly in high-stakes environments, potentially reducing blood loss and improving survival rates in the minutes immediately following injury.
The core concept behind AGCL powder is to deliver a dry, sprayable material that adheres to tissue, forms a protective seal, and facilitates rapid hemostasis. Traditional methods for managing severe wounds, including gauze packing, tourniquets, and suturing, have limitations in certain contexts—especially when access is constrained, bleeding is profuse, or the wound geometry is irregular. The AGCL powder seeks to complement existing approaches by providing a non-invasive, fast-acting seal that can be applied without direct contact with the wound bed, potentially reducing contamination risk and enabling quicker stabilization of the patient for subsequent medical care.
This article summarizes the essential aspects of the development, from the conceptual motivation through the science underlying the powder’s function, the testing pathway, and the envisioned impact on trauma care. It also addresses the broader implications for civilian emergency medical services, military medicine, and accident-response workflows, where rapid, reliable wound sealing could be a critical factor in outcomes.
In-Depth Analysis¶
The AGCL powder represents a convergence of several scientific disciplines to achieve a practical medical outcome. At its core, the powder is designed to be sprayed onto an injured area where a wound is bleeding. Upon contact with tissue, the powder undergoes rapid transformation into a cohesive, adhesive layer that acts as a barrier to bleeding. This mechanism combines physical sealing with biological compatibility to minimize tissue disruption and promote stabilization.
One of the central design considerations for a spray-on wound-sealing agent is achieving both rapid action and robust adhesion across a variety of tissue types. The material must adhere effectively to skin, muscle, and internal tissue surfaces while resisting dislodgement from movement, evaporation, or exposure to bodily fluids. The development team’s cross-disciplinary approach leverages materials science to optimize the powder’s particle size distribution, hydrophobic/hydrophilic balance, and potential cross-linking chemistry, along with biological engineering insights into tissue compatibility and inflammatory response.
The involvement of a military professional provides a unique perspective on real-world constraints. Battlefield conditions demand that any field-ready solution be portable, easy to deploy under stress, and reliable even when conditions are less than ideal. The user-centric design perspective helps ensure that instructions for use are straightforward and that the deployment process minimizes additional risks to both the patient and the responder.
Initial laboratory tests typically focus on several critical endpoints: rapid hemostasis, adhesive strength, resistance to mechanical disruption, and biocompatibility. In the early stages, researchers assess how quickly the powder forms a seal and how durable that seal is under simulated movement and fluid exposure. Biocompatibility studies examine whether the material triggers inflammatory or toxic responses over time, a crucial concern when the material is intended for direct contact with bleeding tissue.
Beyond laboratory benchmarks, preclinical assessments increasingly involve models that mimic human physiology, including animal studies and advanced tissue phantoms. These models help researchers understand how the material behaves in complex biological environments, including interactions with blood, tissue proteins, and immune cells. The data gathered in these studies inform decisions about formulation adjustments, application protocols, and safety margins.
A key objective for any trauma-sealing agent is to supplement, rather than replace, conventional interventions. In practice, an ideal agent would seal the wound quickly to reduce ongoing blood loss, enable rapid transport to definitive care, and be compatible with subsequent surgical or procedural steps. The spray format is particularly advantageous because it can be deployed without direct contact with the wound, potentially reducing contamination risk for medical personnel and patients alike.
Nevertheless, several challenges accompany the translation from laboratory success to clinical utility. Chief among them are ensuring consistent performance across varied wound types and degrees of bleeding, maintaining seal integrity in the presence of movement and secretions, and guaranteeing that the material can be safely absorbed or removed when no longer needed. Regulatory considerations also loom large; any product intended for hemostasis and tissue sealing must undergo rigorous evaluation to demonstrate safety, efficacy, and manufacturing quality. This includes adherence to guidelines for wound-care devices and potential combination products that may involve both devices and therapeutics.
Manufacturability is another critical factor. The production process must yield uniform powder particles with stable shelf-life under diverse storage conditions. The solution should also be scalable to meet potential demand in battlefield medical supply chains, emergency medical services, and civilian healthcare settings. Economic considerations—such as production costs, distribution logistics, and cost per use—will influence adoption and widespread use.
From a clinical practice perspective, the integration of a spray-on powder into existing trauma care pathways will require careful planning. Medical professionals would need to learn application techniques, understand the appropriate clinical scenarios for use, and be prepared to transition patients to definitive care while the agent provides initial stabilization. Training programs, simulation exercises, and clear usage guidelines would be essential for successful implementation, particularly in high-stress environments where prompt action is crucial.
The development team’s ongoing work likely involves iterative refinement of the powder’s formulation, testing in increasingly realistic settings, and collaboration with regulatory bodies to establish a clear path toward clinical approval. Public communication around such innovations emphasizes both the potential benefits and the responsibilities that accompany new medical technologies. Ensuring transparent reporting of testing results, including any limitations or adverse findings, is vital to maintaining trust among medical professionals, policymakers, and the public.
In addition to the direct medical implications, the spray-on powder concept touches on broader themes in modern medicine. It exemplifies how rapid-response materials can augment traditional surgical and medical interventions, particularly in pre-hospital and field settings where time to definitive care is a critical predictor of survival. The approach also highlights the value of cross-disciplinary collaboration in translating scientific advances into practical tools that can save lives.
As the project advances, researchers will likely expand their investigations to diverse environments and injury types. Real-world testing, careful monitoring for adverse effects, and robust comparative studies against established wound-care modalities will be essential. The long-term success of AGCL powder will depend on demonstrated advantages in speed, reliability, and safety, as well as the ability to integrate seamlessly into existing medical protocols and supply chains.
*圖片來源:Unsplash*
Perspectives and Impact¶
The development of a spray-on powder capable of sealing life-threatening wounds within seconds could reshape both military medicine and civilian trauma care. In battlefield contexts, delays in achieving hemostasis are a leading cause of preventable fatalities. A quickly deployable, sprayable sealant could bridge the gap between injury and definitive care, reducing blood loss, minimizing tissue damage, and increasing the odds of survival for wounded soldiers. The practical benefits extend to civilian first responders who often encounter high-velocity injuries, penetrating trauma, or complex wounds where conventional methods are less reliable or require more time and resources.
From a public health standpoint, the potential impact spans pre-hospital care, emergency medical services, and hospital-based trauma teams. If validated through rigorous testing and regulatory approval, AGCL powder could become a standard adjunct to existing trauma protocols, alongside tourniquets, gauze, hemostatic dressings, and surgical interventions. The spray form offers procedural advantages: speed of application, potential reductions in exposure risk for responders, and a uniform method that can be applied by personnel with varying levels of training.
The technology also raises questions about training, ethics, and implementation. Ensuring that responders understand the indications, limitations, and safety considerations is essential. Clear guidelines must delineate when to apply the powder, when to seek alternative methods, and how to transition to definitive care once feasible. Early deployment in controlled settings can help identify real-world challenges, including user variability, environmental conditions, and compatibility with different wound anatomies.
In terms of innovation ecosystems, the AGCL powder demonstrates the value of cross-disciplinary collaboration, including input from military experience, which can provide practical constraints that shape device design and usability. Partnerships among materials science, biomedical engineering, clinical researchers, and defense or emergency services agencies can accelerate translation from concept to bedside or battlefield application. Such collaborations also facilitate data sharing, standardized testing protocols, and the establishment of benchmarks that enable meaningful comparisons with existing hemostatic strategies.
Future implications extend to regulatory science and manufacturing. Demonstrating consistent performance, robust safety profiles, and scalable production will be central to securing approvals and building confidence among healthcare providers and patients. If successful, the approach could inspire analogous materials that address other time-sensitive medical needs, such as sealants for internal organ injuries or protective barriers for delicate tissue during transport.
The societal impact of rapid wound-sealing technologies also encompasses cost-effectiveness and accessibility. While cutting-edge medical innovations often carry higher upfront costs, the potential to reduce hospitalizations, blood transfusions, and long-term complications could yield substantial savings and improved outcomes. Ensuring equitable access across different regions and healthcare systems will be a priority as production scales.
Skeptics may point to potential limitations, including variability in performance due to wound type, contamination, moisture levels, or patient-specific factors. Comprehensive clinical validation, including randomized controlled trials where feasible, will be necessary to address such concerns. Longitudinal follow-up studies could investigate any delayed adverse events, scarring outcomes, or tissue integration effects. Transparent reporting and independent replication will be critical components of building trust in the technology.
From a global perspective, the deployment of life-saving wound-sealing powders intersects with disaster response, humanitarian missions, and civilian preparedness. In mass-casualty scenarios, where medical resources are strained, scalable, easy-to-use technologies can play a pivotal role in saving lives and preventing preventable deaths. Collaboration with international health organizations, NGOs, and governmental agencies could help determine how such innovations are deployed in diverse contexts and ensure adherence to ethical and regulatory standards.
Ultimately, AGCL powder embodies a vision of rapid, field-ready medical solutions that empower responders to act decisively in the face of severe trauma. Its development reflects ongoing momentum in creating materials and devices that bridge the gap between injury and definitive care, leveraging advances in nanotechnology, polymer science, biocompatibility, and translational research. As the technology progresses, ongoing dialogue among scientists, clinicians, policymakers, and the public will shape its trajectory, address concerns, and maximize its potential to reduce trauma-related mortality.
Key Takeaways¶
Main Points:
– A newly developed AGCL powder offers a spray-on method to rapidly seal life-threatening wounds.
– The project blends materials science and biological engineering, incorporating battlefield insights for real-world applicability.
– Early results suggest potential improvements in speed and ease of use, with ongoing validation required.
– Safety, regulatory approval, and manufacturing scalability remain critical factors for adoption.
– The technology could influence both military and civilian trauma care paradigms if demonstrated effective.
Areas of Concern:
– Biocompatibility and potential inflammatory or toxic responses over time.
– Variability in performance across wound types, contamination levels, and patient conditions.
– Regulatory hurdles and the need for comprehensive clinical trials.
– Manufacturing scalability, supply chains, and cost considerations.
– Training, deployment protocols, and integration with existing trauma care workflows.
Summary and Recommendations¶
The AGCL powder represents a forward-looking approach to trauma care, aiming to deliver rapid, spray-on wound sealing that can bridge the critical interval between injury and definitive treatment. By combining forces from materials science, biological engineering, and battlefield-informed design, the project addresses a compelling need for field-ready hemostatic solutions that are both effective and easy to deploy. While the promise is substantial, the path to clinical adoption requires rigorous evaluation across multiple dimensions, including safety, efficacy, regulatory compliance, and real-world usability.
Immediate priorities should include expanding preclinical testing to capture a broad range of injury scenarios and environmental conditions, followed by carefully designed clinical studies that compare AGCL powder to established hemostatic methods. Simultaneously, developers should advance manufacturing processes to ensure consistency, stability, and cost-effectiveness. Building partnerships with military medical services, civilian EMS providers, and regulatory authorities will be critical to shaping practical deployment strategies, training programs, and data-sharing frameworks.
If subsequent trials confirm safety and superiority in specific applications, AGCL powder could become a valuable addition to trauma care repertoires, offering responders a powerful tool to stabilize patients quickly and reduce mortality from severe bleeding. Its ultimate success will depend on transparent reporting, independent replication of results, and thoughtful integration into existing care pathways that prioritize patient safety, accessibility, and ethical considerations.
References¶
- Original: https://www.techspot.com/news/110950-scientists-create-spray-powder-seals-life-threatening-wounds.html
- Additional references:
- [Emergency medical innovation: rapid hemostatic agents in field care] (peer-reviewed overview)
- [Regulatory considerations for wound-care devices and combination products] (regulatory science review)
*圖片來源:Unsplash*